
The catalytic converter is a crucial component of a vehicle’s exhaust system that helps reduce emissions and pollution. Not many car owners know about the importance of catalytic converters or how they work. Its inclusion in the exhaust system of a vehicle is a vital factor in making the car conform to emission standards such as Bharat Stage 6 pollution norms.
The first catalytic converter prototypes were developed in France at the end of the 19th Century when there were only a small number of gasoline cars on the road. Today, almost all modern vehicles come factory fitted with an advanced catalytic converter that keeps emissions in check. From the early days, precious metals were used in catalytic converters, but now we have composite materials and ceramic compounds in honeycomb structures that are extremely efficient in providing clean emissions.
Let’s have a detailed look at what is a catalytic converter and how they work.
What is a Catalytic Converter?
The function of catalytic converter is to control emissions of carbon monoxide and unburned hydrocarbons. Catalytic converters work by converting toxic gases in the exhaust to less harmful pollutants which do not poison the environment. These devices are commonly found on all internal combustion vehicles powered by petrol or diesel engines. They can also be found on electrical generators, forklifts, mining equipment, ships and even wood stoves.
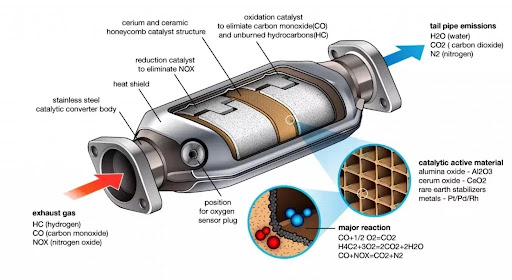
Catalytic converters were made mandatory on all vehicles to comply with stricter pollution norms and environmental protection acts. Some of the polluting gases produced by fossil fuel-powered engines include carbon monoxide, unburned hydrocarbons and nitrogen oxides in addition to lead and Sulphur compounds that are highly toxic. Components in the catalytic converter device transform these poisonous chemicals to inert, clean gases such as carbon dioxide, water vapour and nitrogen dioxide which are found naturally in the atmosphere. Catalytic converter material consists of precious metals such as palladium, platinum, titanium and rhodium to facilitate the conversion of harmful compounds to cleaner emissions. The use of catalytic converters reduces 90% of emissions but their efficiency is impacted by factors such as temperature and the type of fuel used. In addition to these components, a catalytic converter may have particulate filters that can catch ultra-fine particles in exhaust smoke.
Here Checkout What Is An Emission Control System – How It Works?
How Does a Catalytic Converter Work?
The working principle of a catalytic converter is quite simple, based on basic chemical reactions. It is typically a metallic container that houses a ceramic honeycomb structure inside. The ceramic honeycomb is coated with a mix of precious metals such as platinum, palladium and rhodium, which are known as noble metals due to their inert and nonreactive nature. These catalytic converter materials and metals are highly resistant to corrosion, oxidation and harsh acids. When harmful exhaust gases pass through the honeycomb structure, they come in contact with the metal catalysts and chemical reactions convert the toxic pollutants to less harmful compounds.
The catalysts initiate the chemical reaction without being consumed or affecting themselves. The honeycomb structure provides a lot of surface area for the exhaust gases to react with the catalysts. Platinum and rhodium are responsible for reducing nitrogen oxides in exhaust gases, by removing the nitrogen atom and leaving free oxygen atoms that form oxygen gas which is safe to breathe. Meanwhile, platinum and palladium reduce hydrocarbons and carbon monoxide in exhaust gases. Carbon monoxide and oxygen combine to form carbon dioxide and unburned hydrocarbons and oxygen combine to form carbon dioxide and water.
As a result, 90% of exhaust fumes exiting the catalytic converter are breathable and environmentally friendly. Exhaust systems also have oxygen sensors that detect the ratio of fuel to air in the exhaust and adjust the ratio to prevent excessive emissions.
Here Checkout The Signs Of Bad Ignition Coils In An Engine
What Are the Different Types of Catalytic Converters?
There are several main types of catalytic converters depending on the type of materials used in them and whether they are used in petrol on diesel engines.

Two-way catalytic converters:
This is the most common type of catalytic converter used in petrol engines. They are designed to convert harmful carbon monoxide and unburned hydrocarbons to carbon dioxide and water in the exhaust gases.
3 Way Catalytic Converter:
These are also known as closed-loop catalytic converters and are used in petrol engines. Catalytic converter three-way is designed to reduce carbon monoxide, unburned hydrocarbons and nitrogen oxides in exhaust emissions.
Diesel Oxidation Catalyst:
Most commonly found on diesel-powered engines to oxidize particulate matter and hydrocarbons in the exhaust gases.
Lean Nitrogen Oxide Catalyst:
Also used in diesel engines to reduce the nitrogen oxide emissions in lean burn engines.
Selective Catalytic Reduction Converters:
Used in diesel engines to reduce nitrogen oxide by injecting a reducing agent such as urea into the exhaust system.
Particulate Filters:
Are able to catch ultra-fine particles in emissions to increase efficiency to 99%
Read here about Cold Air Intake and its Pros And Cons
How Do You Know When Your Catalytic Converter is Faulty?
Catalytic converters have a life of around 10 years but can vary depending on driving conditions, engine maintenance, vehicle usage and the quality of the catalytic converter. Frequent stop-and-go driving can cause the catalytic converter to heat up and cool rapidly which will shorten its lifespan. The quality of fuel and regular oil changes can increase the life of your catalytic converter. On average, a catalytic converter can last up to 100,000 km without any problems.

Here are some signs that your catalytic converter may be malfunctioning:
Poor Engine Performance: A clogged catalytic converter will restrict the flow of exhaust gases causing a decrease in engine performance, acceleration and top speed.
‘Check engine light’: The onboard diagnostic system consists of sensors on the exhaust system that can detect malfunctions. They will trigger a warning light in the instrument cluster of modern cars.
Increased Fuel Consumption: A faulty catalytic converter will trigger the fuel injector to dump more fuel into the cylinders leading to increased fuel consumption.
Burning Smell From the Exhaust System: A damaged catalytic convert will emit the smell of burning sulphur from the exhaust fumes.
Overheating: A clogged catalytic converter will overheat easily and cause a rise in exhaust gas temperatures.
Reduction in Efficiency: A faulty catalytic converter will not be able to clean exhaust fumes and cause an increase in pollution.
Some ways to keep your catalytic converter working well include avoiding crowded city traffic. Also, use the best quality unleaded fuel and change the engine oil at regular intervals. Highway driving is considered less harmful for the catalytic converter. It is important to note that the symptoms mentioned above can also indicate other underlying problems with the vehicle’s exhaust system. It is recommended to have your vehicle inspected by a qualified mechanic if you suspect a problem with your catalytic converter.
Also Checkout What Is A Fuel Injection System? And It’s Purpose
Conclusion
Catalytic converters play an important role in reducing city pollution by converting harmful emissions into environmentally friendly exhaust gases from internal combustion engines. With regular maintenance and careful driving, you can ensure that the catalytic converter lasts the lifetime of your vehicle and passes emission tests when they are due.
If you found this article/blog on catalytic converters informative and interesting, you may want to explore more of our engaging content on the Carorbis blog such as a Guide On How To Find And Fix Exhaust Leaks In A Car or What Is A Fuel Injection System? And It’s Purpose
Frequently Asked Questions
Q1. What is a Catalytic Converter?
Ans. Catalytic converters are installed in the exhaust system of internal combustion engines to reduce harmful emissions and pollution. It contains a component called a catalyst which is usually made from precious metals such as platinum, palladium, silver and rhodium. These metal catalysts are able to convert toxic carbon monoxide, hydrocarbons and nitrogen oxides into clean carbon dioxide, water and nitrogen. Catalytic converters are placed after the exhaust manifold and before the muffler to reduce harmful emissions by up to 90%. Catalytic converters are a legal requirement on modern vehicles and most cars will not pass an emissions test or conform to BS6 standards without a functional catalytic converter.
Q2. Explain the Working of a Catalytic Converter.
Ans. Exhaust gases, from the result of the combustion of petrol or diesel, flow through the catalytic converter into the atmosphere through the tailpipe. In the process, harmful pollutants are converted into less toxic gases which will not poison the environment. Pollutants such as carbon monoxide, burnt hydrocarbons and nitrogen oxides are eliminated. The catalytic converter consists of a honeycomb structure to increase the surface area of the precious metals in contact with exhaust fumes. More efficient conversion of pollution to cleaner gases occurs with an increased surface area. Catalytic converters can get clogged with oil and burnt fuel over time which can reduce their efficiency. It is important to maintain the exhaust system for optimal functioning.
Q3. What Are the Most Common Catalytic Converter Materials?
Ans. Catalytic converters are generally made from precious metals, ceramics, nanomaterials and a combination of these metals and non-metals. The most common precious metals in use for catalytic converters include platinum, palladium and rhodium. Ceramic materials such as cordierite and aluminium titanate are used to support the structure of precious metals, and they also provide high thermal stability and durability. Other metals used in catalytic converters are stainless steel and titanium for their heat resistance. Nanomaterials such as zeolite, carbon nanotubes and metal oxide nano particles are used to increase the surface area and efficacy of the catalytic reactions.
Q4. What is a Three-Way Catalytic Converter?
Ans. A three-way catalytic converter is installed to reduce three specific pollutants in exhaust gases: carbon monoxide, hydrocarbons and nitrogen oxides. These pollutants are toxic, harmful chemicals to the environment and can cause negative health effects. Three-way catalytic converters reduce emissions of burnt petrol and diesel fuel in internal combustion engines. Three-way catalytic converters are only effective in a specific range of temperatures, so it takes a while for the exhaust system to warm up before it can start working. To circumvent this problem, catalytic converters are placed close to the engine to reach operating temperatures as soon as possible.
Q5. What Are the Different Types of Catalytic Converters?
Ans. There are three main types of catalytic converters used in cars today, namely, oxidation catalysts, three-way catalysts and a combination of these two types.
Oxidation catalysts are able to convert carbon monoxide and hydrocarbons to carbon dioxide and water to prevent pollution. There must be a fresh supply of pure air for this catalytic converter to work well.
Catalytic converter 3 way is the most used type of exhaust system that converts Nitrogen Oxide to Nitrogen in addition to treating carbon monoxide and hydrocarbons.
A combination of these two types of catalytic converters aims to reduce pollution and toxic exhaust gases even more efficiently.
Q6. Is Catalytic Converter Theft Common?
Ans. Catalytic converter theft has been a problem in recent years due to the developed recycling business that uses precious metals such as palladium and platinum. It is often easy to steal catalytic converters because they are located underneath a vehicle, making them a target for thieves. The use of additional security measures such as installing fasteners and bolts can safeguard catalytic converters and prevent thefts. Parking your car in a safe area or in an enclosed parking garage is recommended if possible.
Q7. What Are the Symptoms of a Faulty Catalytic Converter?
Ans. Some common signs of a problematic catalytic converter include reduced engine performance and excessive exhaust smoke. A clogged catalytic converter will restrict exhaust flow causing reduced engine performance and increased consumption of fuel. When the catalytic converter stops working, you will notice thick exhaust smoke and a smell of burning Sulphur. It may trigger the check engine light to illuminate on your instrument cluster due to damage to oxygen sensors in the exhaust system which start to send wrong signals to the fuel injectors, etc.







